 Making a paper airplane in school used to mean trouble. Today it signals a promising discovery in materials science research that could help next-generation technology –like wearable energy storage devices- get off the ground. Researchers at Drexel University and Dalian University of Technology in China have chemically engineered a new, electrically conductive nanomaterial that is flexible enough to fold, but strong enough to support many times its own weight. They believe it can be used to improve electrical energy storage, water filtration and radiofrequency shielding in technology from portable electronics to coaxial cables.
Making a paper airplane in school used to mean trouble. Today it signals a promising discovery in materials science research that could help next-generation technology –like wearable energy storage devices- get off the ground. Researchers at Drexel University and Dalian University of Technology in China have chemically engineered a new, electrically conductive nanomaterial that is flexible enough to fold, but strong enough to support many times its own weight. They believe it can be used to improve electrical energy storage, water filtration and radiofrequency shielding in technology from portable electronics to coaxial cables.
Finding or making a thin material that is useful for holding and disbursing an electric charge and can be contorted into a variety of shapes, is a rarity in the field of materials science. Tensile strength -the strength of the material when it is stretched and compressive strength –its ability to support weight- are valuable characteristics for these materials because, at just a few atoms thick, their utility figures almost entirely on their physical versatility.
Drexel Engineers Improve Strength, Flexibility of Atom-Thick Films
“Take the electrode of the small lithium-ion battery that powers your watch, for example, ideally the conductive material in that electrode would be very small –so you don’t have a bulky watch strapped to your wrist- and hold enough energy to run your watch for a long period of time,” said Michel Barsoum, PhD, Distinguished Professor in the College of Engineering. “But what if we wanted to make the watch’s wristband into the battery? Then we’d still want to use a conductive material that is very thin and can store energy, but it would also need to be flexible enough to bend around your wrist. As you can see, just by changing one physical property of the material –flexibility or tensile strength- we open a new world of possibilities.”
This flexible new material, which the group has identified as a conductive polymer nanocomposite, is the latest expression of the ongoing research in Drexel’s Department of Materials Science and Engineering on a family of composite two-dimensional materials called MXenes.
This development was facilitated by collaboration between research groups of Yury Gogotsi, PhD, Distinguished University and Trustee Chair professor in the College of Engineering at Drexel, and Jieshan Qiu, vice dean for research of the School of Chemical Engineering at Dalian University of Technology in China. Zheng Ling, a doctoral student from Dalian, spent a year at Drexel, spearheading the research that led to the first MXene-polymer composites. The researchat Drexel was funded by grants from the National Science Foundation and the U.S. Department of Energy.
The Drexel team has been diligently examining MXenes like a paleontologist carefully brushing away sediment to unearth a scientific treasure. Since inventing the layered carbide material in 2011 the engineers are finding ways to take advantage of its chemical and physical makeup to create conductive materials with a variety of other useful properties.
One of the most successful ways they’ve developed to help MXenes express their array of abilities is a process, called intercalation, which involves adding various chemical compounds in a liquid form. This allows the molecules to settle between the layers of the MXene and, in doing so, alter its physical and chemical properties. Some of the first, and most impressive of their findings, showed that MXenes have a great potential for energy storage.

“The uniqueness of MXenes comes from the fact that their surface is full of functional groups, such as hydroxyl, leading to a tight bonding between the MXene flakes and polymer molecules, while preserving the metallic conductivity of nanometer-thin carbide layers. This leads to a nanocomposite with a unique combination of properties,” Gogotsi said.
The results of both sets of MXene testing were recently published in the Proceedings of the National Academy of Sciences. In the paper Flexible and conductive MXene films and nanocomposites with high capacitance, the researchers report that the material exhibits increased ability to store charge over the original MXene; and 300-400 percent improvement in strength.
“We have shown that the volumetric capacitance of an MXene-polymer nanocomposite can be much higher compared to conventional carbon-based electrodes or even graphene,” said Chang Ren, Gogotsi’s doctoral student at Drexel. “When mixing MXene with PVA containing some electrolyte salt, the polymer plays the role of electrolyte, but it also improves the capacitance because it slightly enlarges the interlayer space between MXene flakes, allowing ions to penetrate deep into the electrode; ions also stay trapped near the MXene flakes by the polymer. With these conductive electrodes and no liquid electrolyte, we can eventually eliminate metal current collectors and make lighter and thinner supercapacitors.”
Though just a few atoms thick, the MXene-polymer nanocomposite material shows exceptional strength -especially when rolled into a tube.
The testing also revealed hydrophilic properties of the nanocomposite, which means that it could have uses in water treatment systems, such as membrane for water purification or desalinization, because it remains stable in water without breaking up or dissolving.
In addition, because the material is extremely flexible, it can be rolled into a tube, which early tests have indicated only serves to increase its mechanical strength. These characteristics mark the trail heads of a variety of paths for research on this nanocomposite material for applications from flexible armor to aerospace components. The next step for the group will be to examine how varying ratios of MXene and polymer will affect the properties of the resulting nanocomposite and also exploring other MXenes and stronger and tougher polymers for structural applications.
Source: www.drexel.edu
RELATED ARTICLES:
Flexible and conductive MXene films and nanocomposites with high capacitance
 Two-dimensional transition metal carbides (MXenes) offer a quite unique combination of excellent mechanical properties, hydrophilic surfaces, and metallic conductivity.
Two-dimensional transition metal carbides (MXenes) offer a quite unique combination of excellent mechanical properties, hydrophilic surfaces, and metallic conductivity.
In this first report on MXene composites of any kind, researchers show that adding polymer binders/spacers between atomically thin MXenes layers or reinforcing polymers with MXenes results in composite films that have excellent flexibility, good tensile and compressive strengths, and electrical conductivity that can be adjusted over a wide range.




 Our collaborative work on porous Ti₃AlC₂ MAX phase for efficient Ti₃C₂Tₓ MXene synthesis has been ranked among the Top 10 most cited papers in the International Journal of Applied Ceramic Technology (IJACT).
Our collaborative work on porous Ti₃AlC₂ MAX phase for efficient Ti₃C₂Tₓ MXene synthesis has been ranked among the Top 10 most cited papers in the International Journal of Applied Ceramic Technology (IJACT). We highly recommend checking out new important paper: “Critical Assessment of Intrinsic Antibacterial Properties and Photothermal Therapy Potential of MXene Nanosheets.” Along with the key findings, we’re also excited to share the Supplementary Cover Art — it beautifully illustrates our vision of MXene-based targeted complexes that can eliminate bacteria via photothermal conversion under near-infrared irradiation.
We highly recommend checking out new important paper: “Critical Assessment of Intrinsic Antibacterial Properties and Photothermal Therapy Potential of MXene Nanosheets.” Along with the key findings, we’re also excited to share the Supplementary Cover Art — it beautifully illustrates our vision of MXene-based targeted complexes that can eliminate bacteria via photothermal conversion under near-infrared irradiation.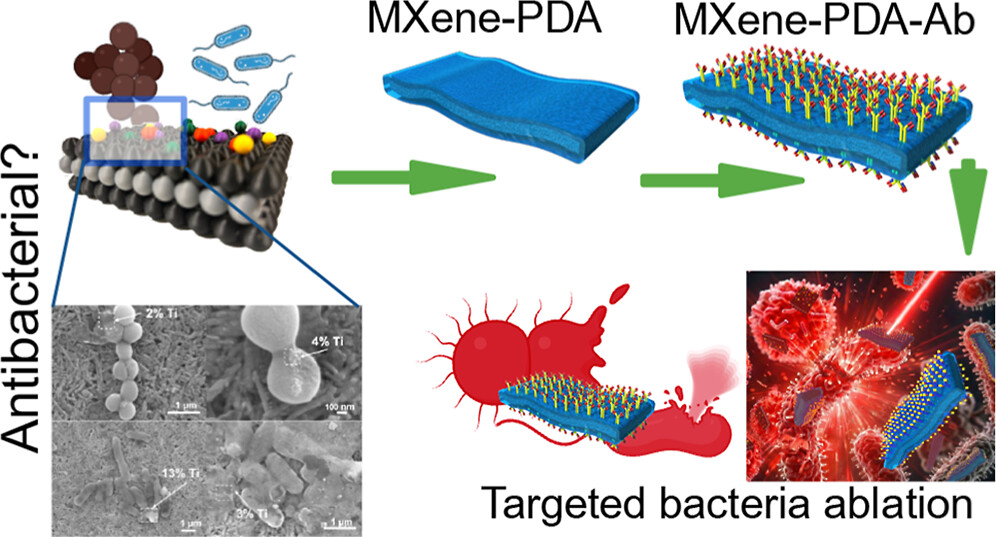 Do MXene nanosheets possess intrinsic antibacterial activity? A systematic study of high-quality Ti-, V-, and Nb-based MXenes reveals negligible inherent antimicrobial effects while highlighting their strong potential for targeted photothermal antibacterial therapy.
Do MXene nanosheets possess intrinsic antibacterial activity? A systematic study of high-quality Ti-, V-, and Nb-based MXenes reveals negligible inherent antimicrobial effects while highlighting their strong potential for targeted photothermal antibacterial therapy. Highlights
Highlights We are excited to share that our Carbon-Ukraine (Y-Carbon LLC) company participated in the I2DM Summit and Expo 2025 at Khalifa University in Abu-Dhabi! Huge thanks to Research & Innovation Center for Graphene and 2D Materials (RIC2D) for hosting such a high-level event.It was an incredible opportunity to meet brilliant researchers and innovators working on the next generation of 2D materials. The insights and energy from the summit will definitely drive new ideas in our own development.
We are excited to share that our Carbon-Ukraine (Y-Carbon LLC) company participated in the I2DM Summit and Expo 2025 at Khalifa University in Abu-Dhabi! Huge thanks to Research & Innovation Center for Graphene and 2D Materials (RIC2D) for hosting such a high-level event.It was an incredible opportunity to meet brilliant researchers and innovators working on the next generation of 2D materials. The insights and energy from the summit will definitely drive new ideas in our own development. Carbon-Ukraine team had the unique opportunity to visit XPANCEO - a Dubai-based deep tech startup company that is developing the first smart contact lenses with AR vision and health monitoring features, working on truly cutting-edge developments.
Carbon-Ukraine team had the unique opportunity to visit XPANCEO - a Dubai-based deep tech startup company that is developing the first smart contact lenses with AR vision and health monitoring features, working on truly cutting-edge developments. Our Carbon-Ukraine team (Y-Carbon LLC) are thrilled to start a new RIC2D project MX-Innovation in collaboration with Drexel University Yury Gogotsi and Khalifa University! Amazing lab tours to project collaborators from Khalifa University, great discussions, strong networking, and a wonderful platform for future collaboration.
Our Carbon-Ukraine team (Y-Carbon LLC) are thrilled to start a new RIC2D project MX-Innovation in collaboration with Drexel University Yury Gogotsi and Khalifa University! Amazing lab tours to project collaborators from Khalifa University, great discussions, strong networking, and a wonderful platform for future collaboration.
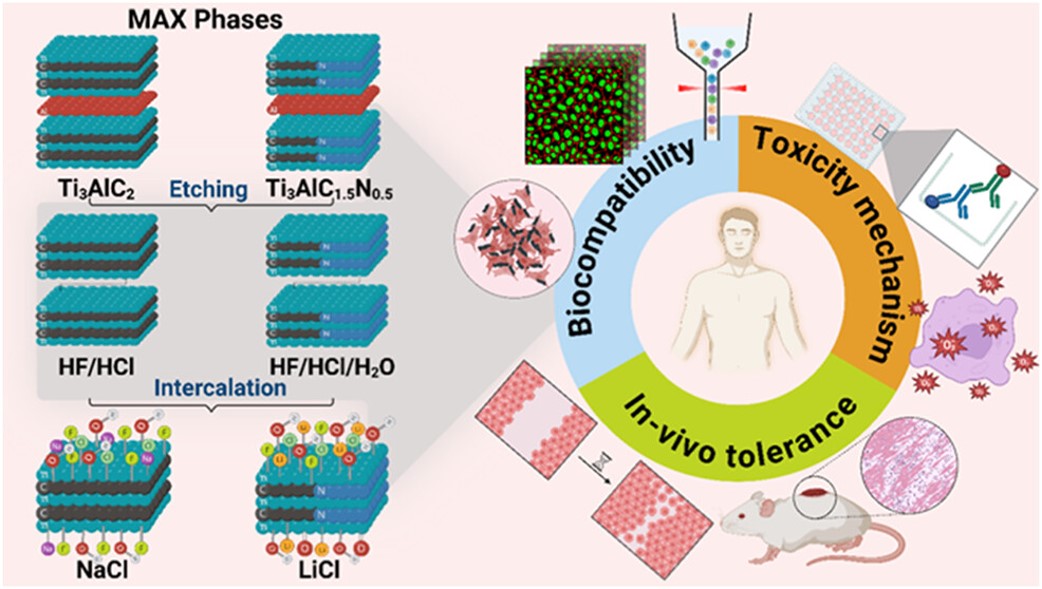 MXenes potential applications include sensors, wound healing materials, and drug delivery systems. A recent study explored how different synthesis methods affect the safety and performance of MXenes. By comparing etching conditions and intercalation strategies, researchers discovered that fine-tuning the surface chemistry of MXenes plays a crucial role in improving biocompatibility. These results provide practical guidelines for developing safer MXenes and bring the field one step closer to real biomedical applications.
MXenes potential applications include sensors, wound healing materials, and drug delivery systems. A recent study explored how different synthesis methods affect the safety and performance of MXenes. By comparing etching conditions and intercalation strategies, researchers discovered that fine-tuning the surface chemistry of MXenes plays a crucial role in improving biocompatibility. These results provide practical guidelines for developing safer MXenes and bring the field one step closer to real biomedical applications.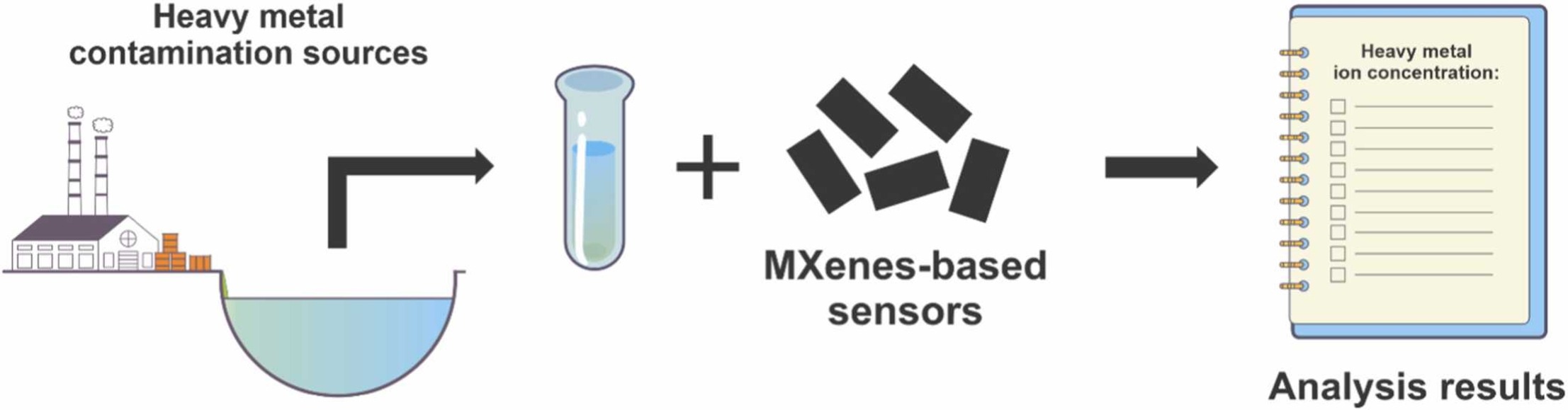 An excellent review highlighting how MXene-based sensors can help tackle one of today’s pressing environmental challenges — heavy metal contamination. Excited to see such impactful work moving the field of environmental monitoring and sensor technology forward!
An excellent review highlighting how MXene-based sensors can help tackle one of today’s pressing environmental challenges — heavy metal contamination. Excited to see such impactful work moving the field of environmental monitoring and sensor technology forward!
 Carbon-Ukraine team was truly delighted to take part in the kickoff meeting of the ATHENA Project (Advanced Digital Engineering Methods to Design MXene-based Nanocomposites for Electro-Magnetic Interference Shielding in Space), supported by NATO through the Science for Peace and Security Programme.
Carbon-Ukraine team was truly delighted to take part in the kickoff meeting of the ATHENA Project (Advanced Digital Engineering Methods to Design MXene-based Nanocomposites for Electro-Magnetic Interference Shielding in Space), supported by NATO through the Science for Peace and Security Programme. Exellent news, our joint patent application with Drexel University on highly porous MAX phase precursor for MXene synthesis published. Congratulations and thanks to all team involved!
Exellent news, our joint patent application with Drexel University on highly porous MAX phase precursor for MXene synthesis published. Congratulations and thanks to all team involved! Our team was very delighted to take part in International Symposium "The MXene Frontier: Transformative Nanomaterials Shaping the Future" – the largest MXene event in Europe this year!
Our team was very delighted to take part in International Symposium "The MXene Frontier: Transformative Nanomaterials Shaping the Future" – the largest MXene event in Europe this year!  Last Call! Have you submitted your abstract for IEEE NAP-2025 yet? Join us at the International Symposium on "The MXene Frontier: Transformative Nanomaterials Shaping the Future" – the largest MXene-focused conference in Europe this year! Final Submission Deadline: May 15, 2025. Don’t miss this exclusive opportunity to showcase your research and engage with world leaders in the MXene field!
Last Call! Have you submitted your abstract for IEEE NAP-2025 yet? Join us at the International Symposium on "The MXene Frontier: Transformative Nanomaterials Shaping the Future" – the largest MXene-focused conference in Europe this year! Final Submission Deadline: May 15, 2025. Don’t miss this exclusive opportunity to showcase your research and engage with world leaders in the MXene field!